Reduce waste and cut down on the number of touchpoints in surgery preparation.
Fagron Sterile Services US (FSS) offers pre-filled, ready-to-administer critical care solutions prepared in cGMP-compliant, DEA- and FDA-registered 503B outsourcing facilities inspected against 21 CFR 210 and 211 standards.
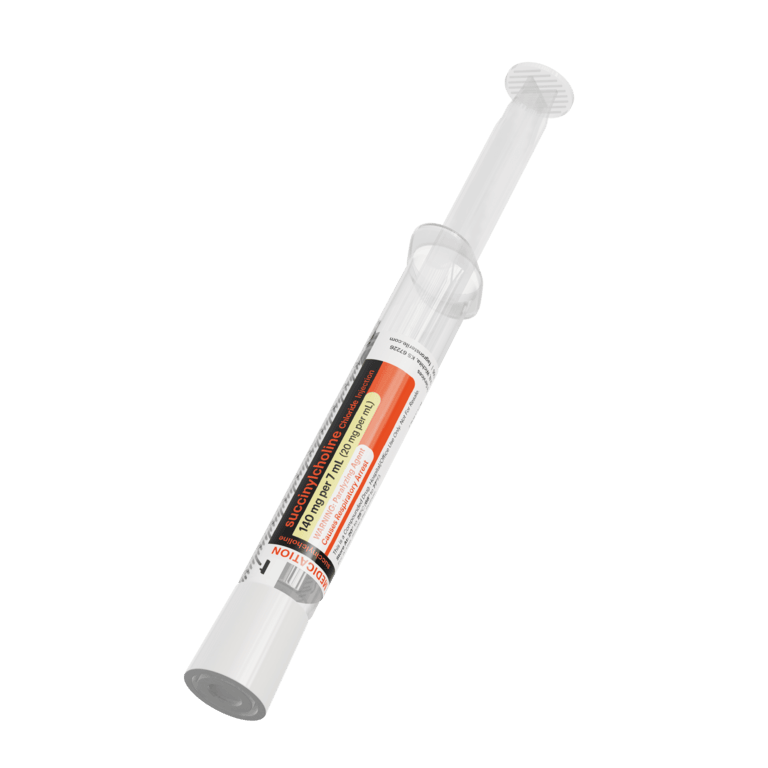
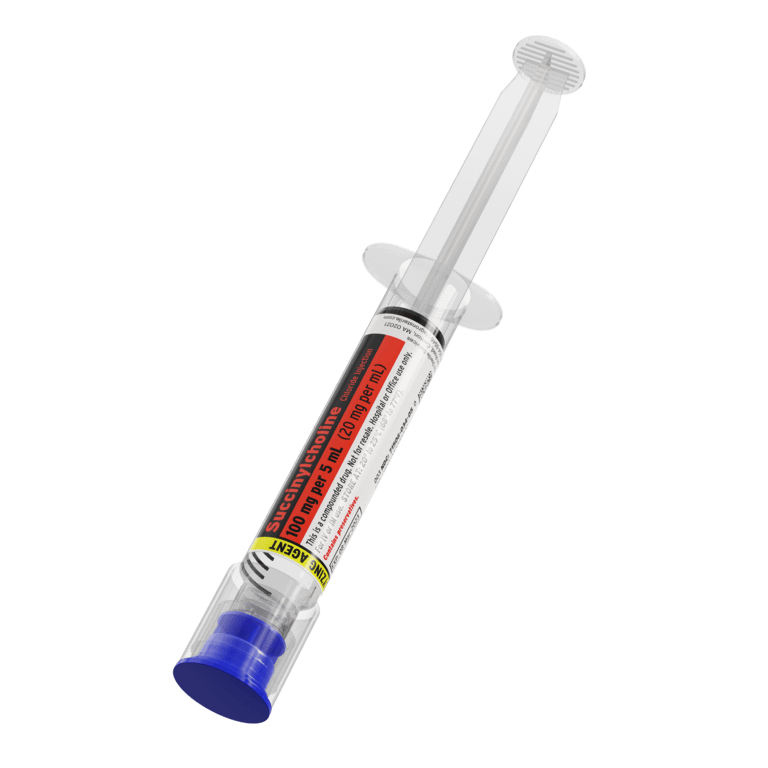
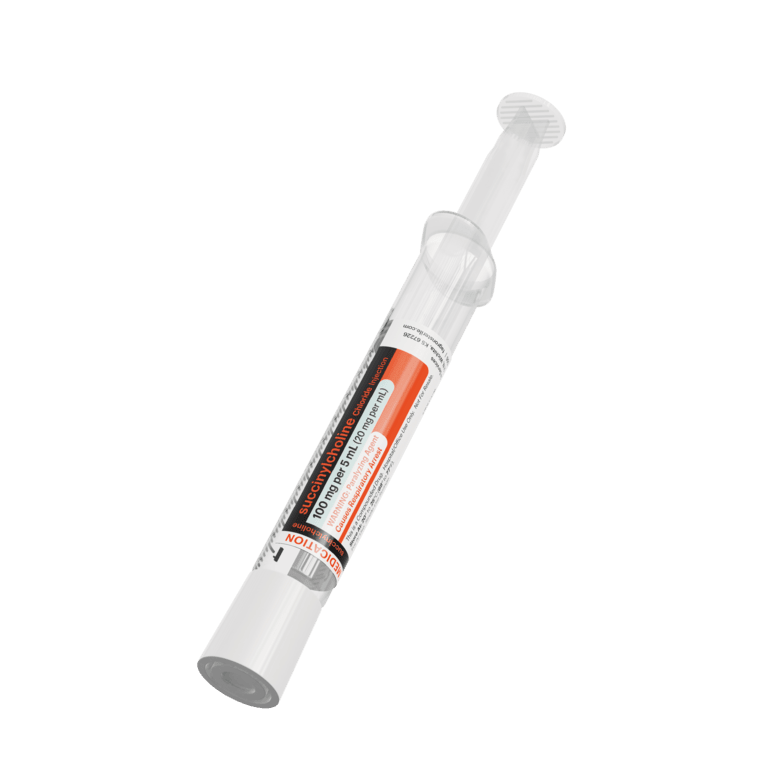
Our advanced label designs, developed in collaboration with customers, align with ISMP and FDA guidance, USP standards, and ASTM color standards to help reduce medication errors. These features support differentiation of look-alike/sound-alike drugs, multiple strengths, and similar containers—enhancing patient safety


503B outsourcing Critical Care medication
Time and accuracy matter in the critical care setting, which is why every ready-to-administer Critical Care syringe and IV bag from Fagron Sterile Services is thoroughly tested. No syringe is released from quarantine until all testing for sterility, particulate matter, and potency is complete and approved by our Quality team.
All Critical Care syringes and IV bags are clearly labeled, adhering to ASTM, DQSA, and ISMP medication and anesthesia labeling standards. This includes TALLman lettering, presentation specifications, barcodes, expiration dating, and storage requirements and helping you mitigate medication errors.
Critical Care solutions are produced with cGMP (21 CFR 210 & 211) compliant operations in our state-of-the-art 503B outsourcing facilities. As an industry leader, FSS has built trusted customer partnerships built around transparency. FSS unique site visit program supports accessibility with virtual and in-person options and welcomes current and prospective customers to experience our state-of-the-art operations first-hand.
Talk with your dedicated Account Representative today to learn more about our Instant Ship program and RFID enabled OR syringes.
Advanced labeling
All labels clearly display the most important product information. Patient safety is paramount, so Fagron Sterile Services US adheres to all ASTM, DQSA, and ISMP medication and anesthesia labeling standards to improve visualization of the most critical elements: Presentation Specifications, Route of Administration, Storage Requirements, TALLman Lettering, Expiration Dating, and Barcode.

Compounding Flexibility
Every facility has different needs. To serve you and the patients who depend on you better, Fagron Sterile Services US proudly offers reliable FDA and DEA inspected capabilities in Sterile-to-Sterile, API-to-Sterile and Biologic compounding. Come see for yourself and schedule a site visit today.

Vertical integration
As part of the only vertically integrated pharmaceutical company dedicated to sterile compounding, our 503B facilities drive outsourcing solutions on a comprehensive level across the continuum of care. Learn more about our affiliate companies today.

Examples of Critical Care products & services
Fagron Sterile Services carries a broad product line of presentations for the most commonly-used Critical Care Solutions including: Neuromuscular Blocking Agents (NMB): Succinylcholine and Rocuronium, and NMB Reversal: Neostigmine.
Speak with your personal representative today
Service is in our name and we can’t wait to partner with your patient-focused healthcare facility. Schedule a free consultation today to discuss your 503B outsourcing needs.

Globally and vertically integrated operations
Fagron’s 4k+ employees serve 30+ countries from 70+ facilities
Proactively supporting supply chain resiliency
Fagron’s global team of 100 supply chain experts leverage 3k+ suppliers
Diverse portfolio across the continuum of care
FSS supports patient care from the delivery room to the O.R.
including clinics